Identifizierung von neuen RubisCOs aus metagenomischen Fosmid-Bibliotheken durch einen aktivitätsbasierten Screeningansatz
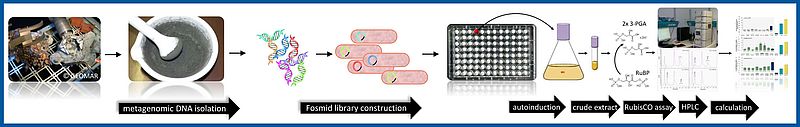
RubisCO (Ribulose-1,5-Bisphosphat-Carboxylase/Oxygenase) ist ein Schlüsselenzym des Calvin-Benson-Bassham-Zyklus, der für die Fixierung von etwa 200 Gigatonnen CO2 pro Jahr verantwortlich ist. In diesem Zyklus vermittelt RubisCO den entscheidenden CO2-Fixierungsschritt, indem es CO2 an Rubp (Ribulose-1,5-bisphosphat) bindet und dabei 3-D-Phosphoglycerat (3-PGA) produziert. Von den vier derzeit bekannten RubisCO-Typen haben nur die Typen I, II und III die klassischen RuBP-abhängigen Carboxylase Aktivitäten. Im Rahmen dieses Projektes haben wir einen aktivitätsbasierten Screen entwickelt, um Enzyme mit RuBP-umwandelnden Eigenschaften identifizieren zu können ohne dabei auf die Kultivierbarkeit des ursprünglichen Wirtsbakteriums angewiesen zu sein. Die Metagenombanken wurden mit Probenmaterial hydrothermalen Ursprungs hergestellt. Wir haben 12.500 Fosmid-Klone gescreent und konnten vierzig aktive RubisCO-Enzyme finden. Sie ähneln Typ I und Typ II RubisCO aus Alpha-, Beta- oder Gammaproteobakterien und sogar Typ III RubisCOs aus Archaeen konnten wir detektieren.
Insbesondere ein metagenomisches Fragment wies sehr interessante Eigenschaften auf: Die exprimierten Proteine wandeln Rubp innerhalb von Sekunden um und die entsprechende DNA-Sequenz ähnelt keinem bekannten RubisCO-Typ. Die Arbeiten an diesem Fragment und den entsprechenden Genprodukten werden momentan weitergeführt.
Beteiligte Mitarbeiter
Zugehörige Puplikationen
Böhnke, S. & Perner, M. (2019)
Seeking active RubisCOs from the currently uncultured microbial majority colonizing deep-sea hydrothermal vent environments. ISME Journal
DOI: 10.1038/s41396-019-0439-3
Böhnke, S. & Perner, M. (2017)
Unraveling RubisCO Form I and Form II Regulation in an Uncultured Organism from a Deep-Sea Hydrothermal Vent via Metagenomic and Mutagenesis studies. Frontiers in Microbiology DOI: 10.3389/fmicb.2017.01303.
Böhnke, S. & Perner, M. (2015)
A function-based screen for seeking RubisCO active clones from metagenomes: novel enzymes influencing RubisCO activity. ISME Journal 9 (3) 735-745. DOI:10.1038/ismej.2014.163
Vorherige Finanzierungen
Dieses Projekt wurde von der Deutschen Forschungsgemeinschaft (DFG) finanziert (2013-2017). Die Exzellenzinitiative Hamburg (LEXI), die BWF und die Joachim Herz Stiftung finanzierten die Graduiertenschule C1-Ressourcen- und Energiemanagement, aus der dieses Projekt finanziert wurde (2009-2013).